Product Information
COVID-19 is a global pandemic caused by SARS-CoV-2, a new coronavirus strain sharing similar structures with other strains in the coronavirus family including SARS, MERS, NL63, 229E, OC43 and HKU1. SARS-CoV-2 is a 30Kb RNA virus encoding 29 structural and non-structure proteins with different functions. SARS-CoV-2 infection initiates production of antibodies against different antigenic epitopes, but only antibodies against the receptor binding domain (RBD) of the Spike protein (S) are protective against viral infection. It is important to identify antibodies against different proteins and their cross-reactivity with antigens from other coronavirus strains. On the other hand, the severity of COVID-19 has been found to be correlated with the level of autoantibodies against self-proteins, for example, the COVID-19 patients with high level of anti-IFNa autoantibody tend to have severe clinical symptoms. Therefore, comprehensive measurement of antibodies against a broad range of self-antigens and viral antigens is critical in COVID-19 research.
GeneCopoeia has developed a new comprehensive antigen protein array that contains 120 antigens, including 21 SARS-CoV-2 and other viral antigens, 42 human cytokines and chemokines, and 57 human self-antigens. This panel can be used for quantitative measurement of various antibody subtypes (e.g. IgG, IgM, IgA) against a broad range of antigens in COVID-19 patients and for evaluation of immune responses to viral infection and vaccination.
In addition to premade arrays, arrays containing customized sets of antigens are available, as well as array processing kits, array profiling services and data analysis. To order premade or custom arrays, please contact us.
GeneCopoeia’s
OmicsArray™ Coronavirus infection-associated Autoimmune Antigen Array is part of the GeneCopoeia OmicsArray™ Antigen Microarray family.
Reference
- Bastard, Paul et al. “Autoantibodies against type I IFNs in patients with life-threatening COVID-19.” Science (New York, N.Y.) vol. 370,6515 (2020): eabd4585. doi:10.1126/science.abd4585
- Choy, Kay Weng. “SARS-CoV-2 serological cross-reactivity with autoantibodies.” The Lancet. Rheumatology vol. 3,1 (2021): e15. doi:10.1016/S2665-9913(20)30358-1
- de Assis, Rafael R et al. “Analysis of SARS-CoV-2 antibodies in COVID-19 convalescent blood using a coronavirus antigen microarray.” Nature communications vol. 12,1 6. 4 Jan. 2021, doi:10.1038/s41467-020-20095-2
- Hou, Xin et al. “Serum Protein Profiling Reveals a Landscape of Inflammation and Immune Signaling in Early-stage COVID-19 Infection.” Molecular & cellular proteomics : MCP vol. 19,11 (2020): 1749-1759. doi:10.1074/mcp.RP120.002128
This array is ideally suited for the following applications for use in clinical and basic research studies:
- Detection of virus-specific antibodies against different antigens of SARS-CoV-2, other coronavirus strains, influenza virus, and respiratory syncitial virus (RSV).
- Detection of autoantibodies against a broad range of human cytokines and chemokines, which is valuable for evaluating the relationship between anti-cytokine antibodies and COVID-19 clinical phenotypes.
- Evaluation of how COVID-19 infection will enhance the body’s autoimmune reaction to various autoantigens and/or the interaction between autoantibodies and COVID-19 antibodies.
- Monitoring the body’s immune reactivity to COVID-19 vaccination, by ensuring the antibody reactivities induced by COVID-19 vaccination and detecting the protective antibodies against RBD of spike antigens.

Figure 1. Arrangement of GeneCopoeia’s Coronavirus-associated Autoimmunity Microarray. Each slide contains 16 arrays. Each array can detect 120 antigen-specific antibodies. The array is highly sensitive for a broad range of sample types including blood (5ul serum or plasma), saliva, sputum, spinal fluid, nasal swabs, tears, etc., and simultaneously detects multiple antibody types (e.g. IgG, IgM, IgA) or subclasses. Array contents: 57 autoantigens, 42 cytokines, 21 virus antigens, and 8 internal controls
The listed chip prices apply to academic customers. For custom arrays and services pricing, please contact us.
Custom services
GeneCopoeia offers custom antigen microarray services in the following areas:
- Custom array printing. GeneCopoeia will create custom antigen microarrays built to your specifications.
- Sample processing. Send us your blood, plasma, tissue, or other biological sample and we will prepare it for processing and incubation with any of our premade antigen microarrays or custom-built antigen microarrays for autoantibody profiling and other applications. For information on sample types to submit, consult the FAQ
- Data analysis. Once samples are processed and incubated with an antigen microarray, we will analyze the raw data. The standard analysis service includes: 1) An Excel file of the Net Signal Intensity (NSI) for each antigen on the array, normalized to internal controls; and 2) a heat map
Additional analysis services, including proteomic analysis, pathway analysis, and more, are also available.
To inquire about custom antigen microarray products and services, please fill out our custom quote request form.
Technology overview
Advantages of OmicsArray™ Antigen Microarrays
- Largest collection of pre-made whole-protein antigen microarrays on the market.
- Largest number of whole-protein antigens specifically focused on autoimmune diseae research.
- Best combination of number of antigens per array (up to 120) with number of samples that can be processed per slide (up to 15).
Technology overview
GeneCopoeia’s OmicsArray™ antigen microarrays contain up to 120 purified proteins spotted onto nitrocellulose filters, which are adhered to glass slides. In addition, 8 spots are included for normalization. Each slide carries 16 identical arrays, and so can be used to process up to 15 samples simultaneously as well as a negative control. As little as 1 ul serum or 50 ul of other bio fluids are needed for each sample.
As shown in Figure 1, arrays are incubated with patient samples, and any antibodies in the samples bind to their cognate antigens on the array. The arrays are washed to remove unbound antibodies and other proteins, then co-incubated with Cy3- and Cy5-labeled secondary antibodies. The dual labeling strategy is intended to distinguish between immunoglobulin (Ig) subtypes present within samples. For example. a Cy3-labeled anti-IgG secondary antibody is used to detect IgG antibodies, and a Cy5-labeled anti-IgM secondary antibody is used to detect IgM antibodies. Fluorophore-labeled secondary antibodies are available for detecting IgA, IgD, IgE, IgG and IgM immunoglubulins, as well as IgG subclasses IgG1, IgG2, IgG3, and IgG4.
After washing to remove unbound secondary antibodies, signals are detected using a microarray scanner (e.g., GenePix® 4000B, InnoScan 710, or equivalents). The raw data is then be analyzed using GenePix® Pro 7.0, or Mapix software.
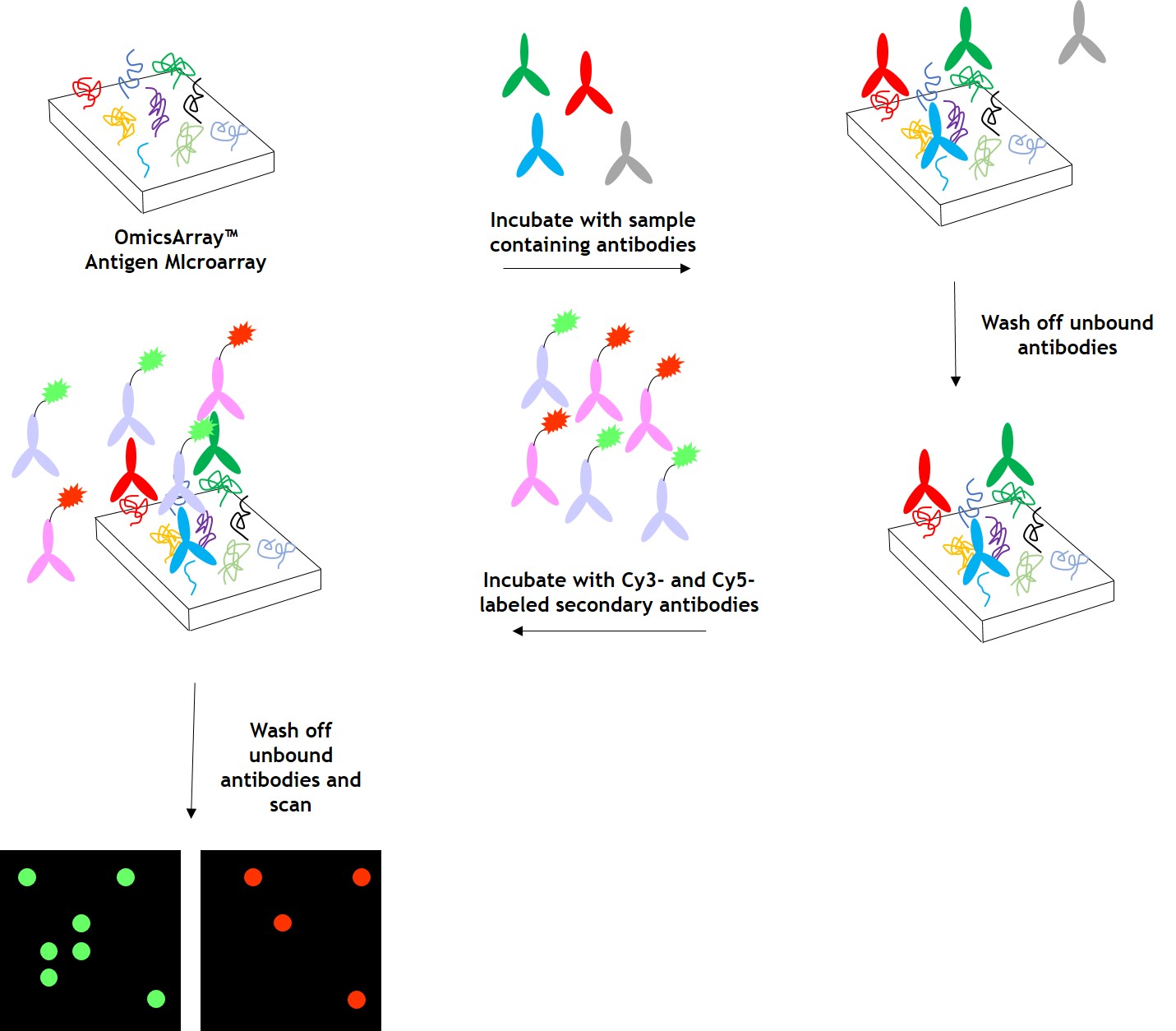
Figure 1. Workflow for detection of antibodies in samples using GeneCopoeia’s OmicsArray™ antigen microarrays.
Data analysis
GeneCopoeia provides data analysis as part of its custom antigen microarray services. After array scanning, raw data are collected and analyzed using GenePix® 7.0 software. The standard service includes data normalization to include fold changes of, for example, disease vs. healthy control, and a heat map showing each antigen ranked by level of fold change. Our service undergoes the following steps:
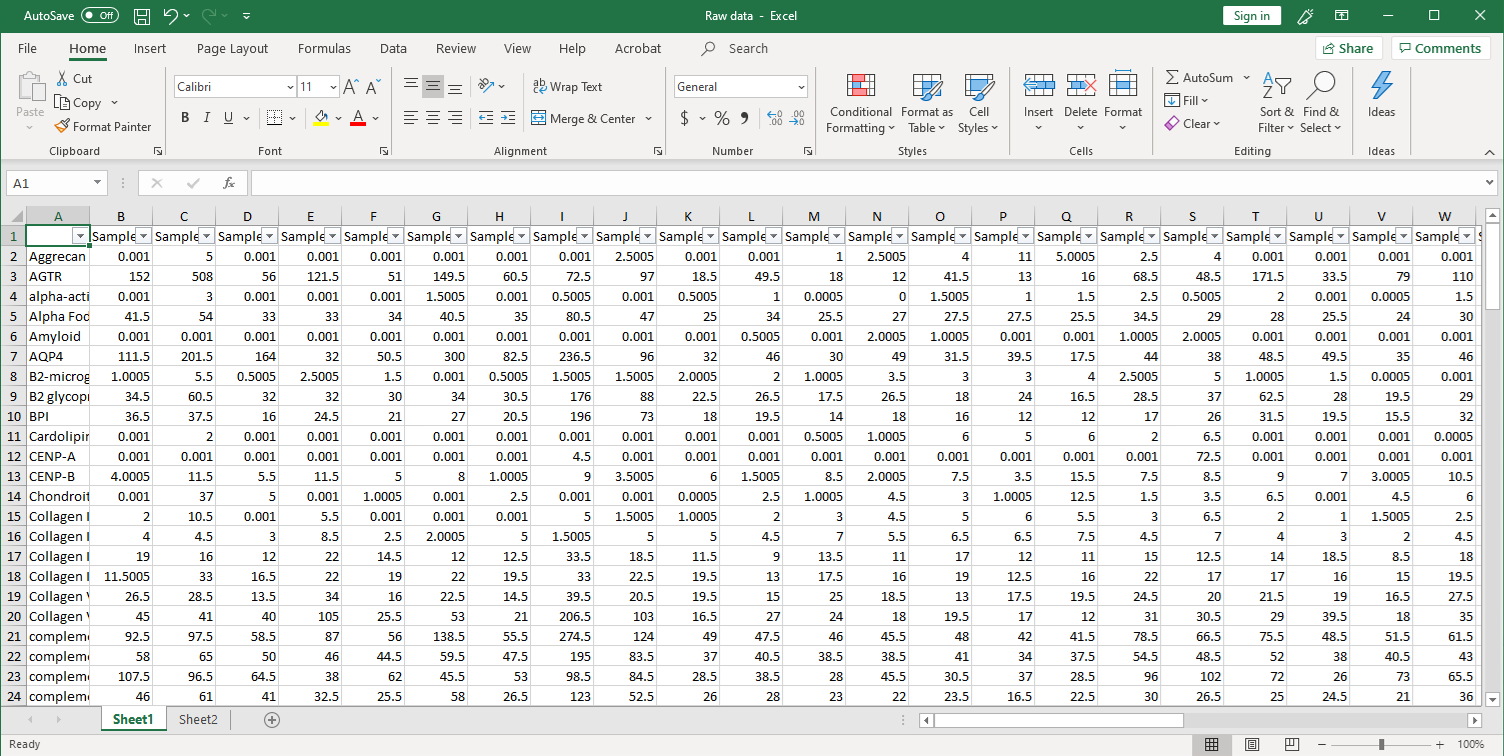
1. Collection of raw data. After scanning, raw signal intensities are collected in an Excel file, as shown below.
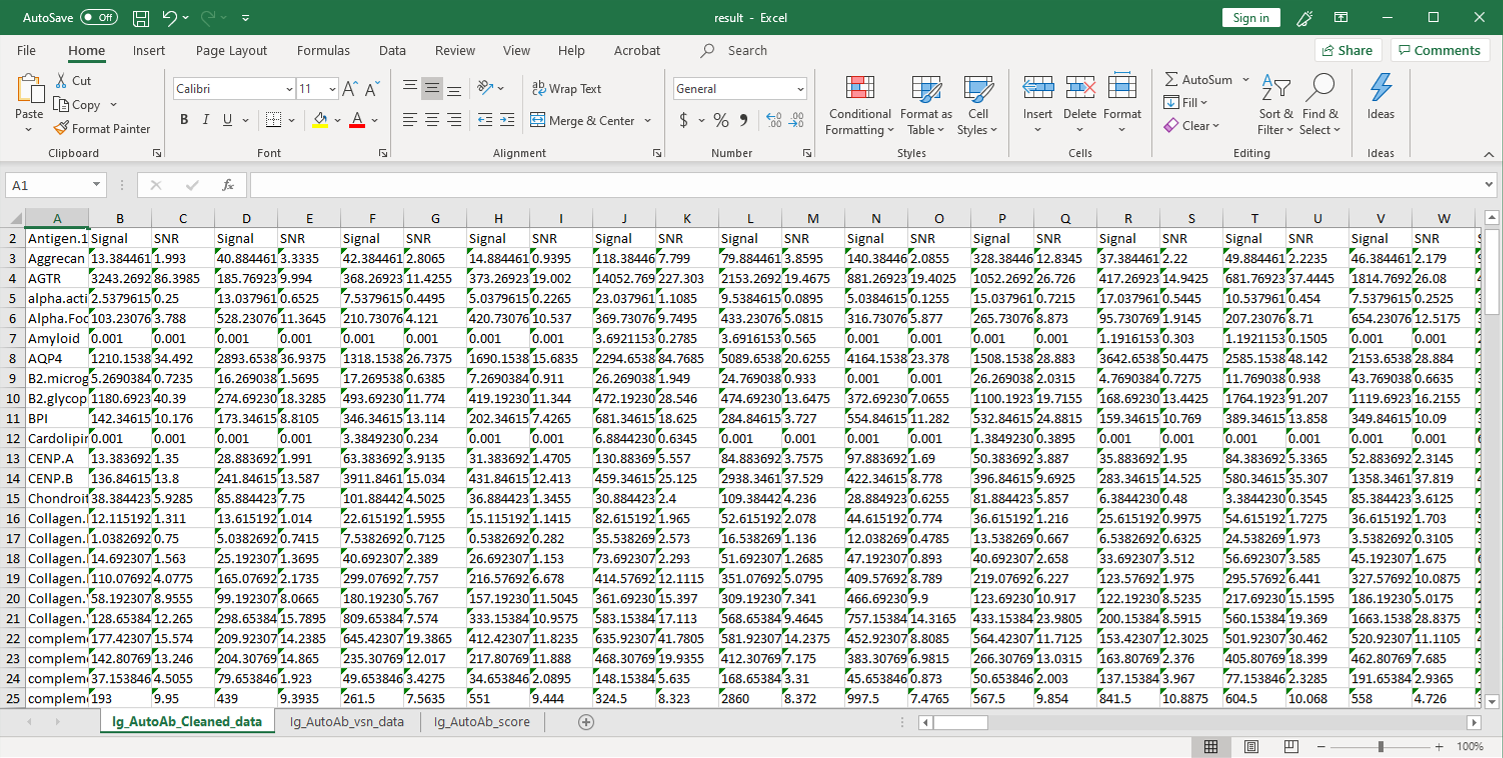
2. Next, the raw data are normalized to controls on the array, and the Net Signal Intensity (NSI) values, as well as the signal-to-noise ratios (SNRs) are tabulated in an Excel file,
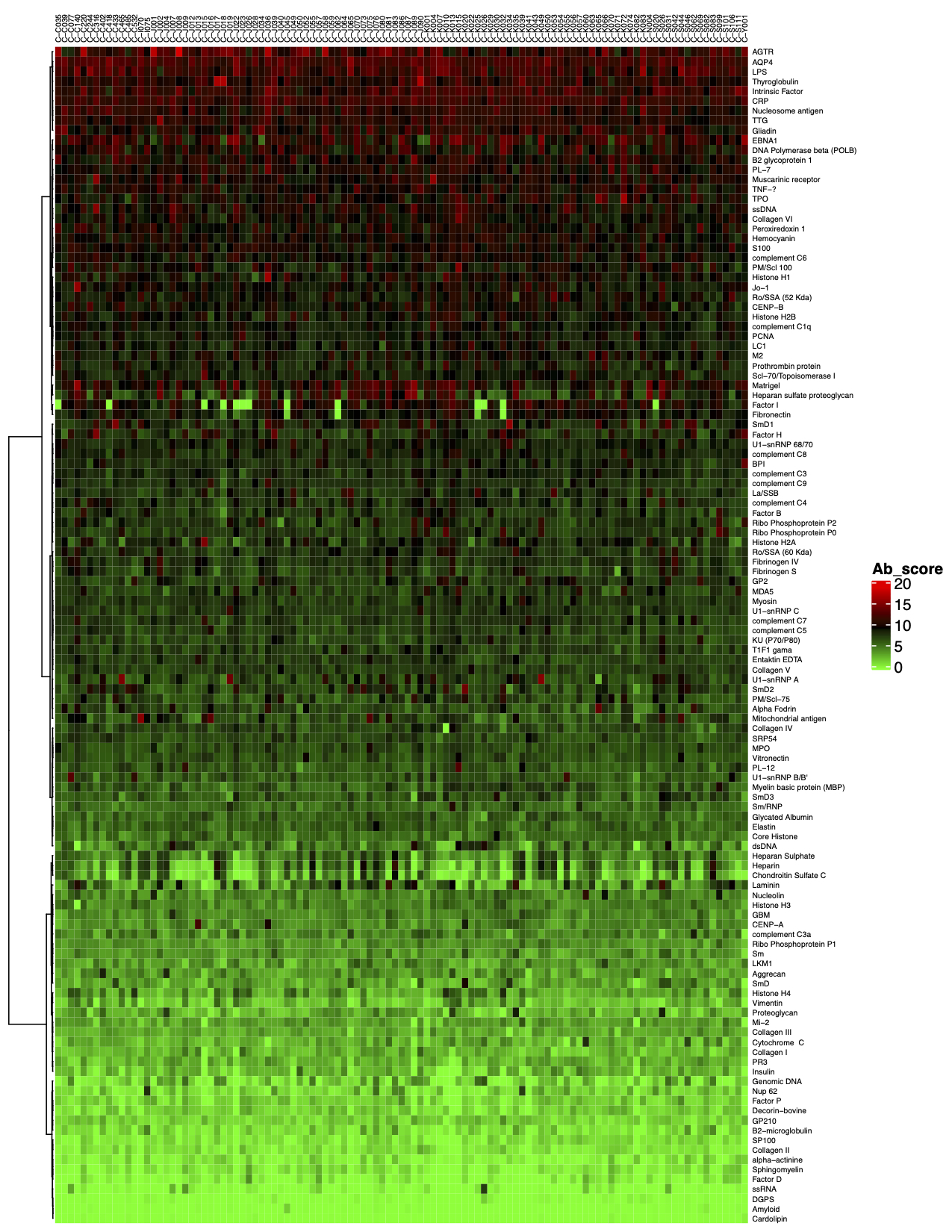
3. The final step of the standard analysis package is to determine the “Antibody score” which is the relative enrichment of a given antibody in a sample. The antibody score is determined numerically and displayed in a heat map. A higher score (shifted toward red on the heat map) suggests a stronger antibody-antigen interaction, as shown in the following example:
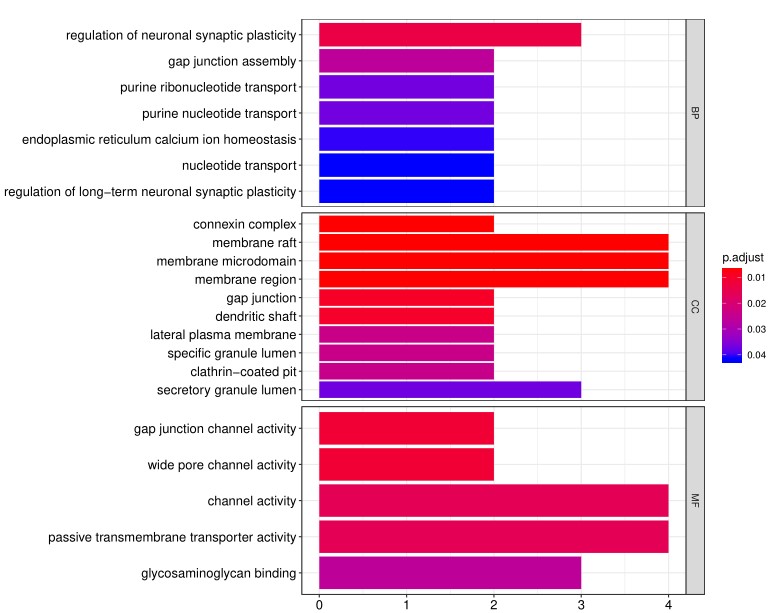
4. In addition to the standard data analysis package, customers can also choose custom data analysis services. One such service is to classify positive antigens on the array using Gene Ontology (GO) analysis, which classifies genes and proteins based on known biolgical functions. An example of the readout from GO analysis for molecular function is shown below:
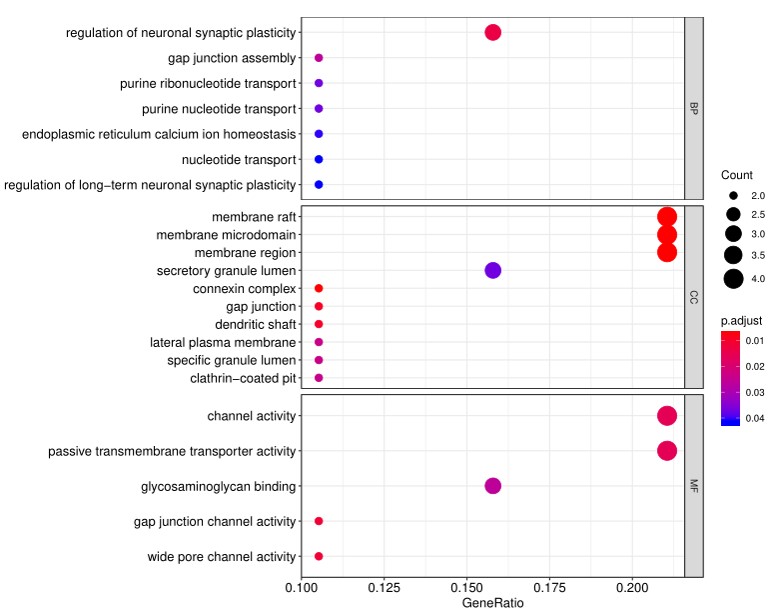
Further, GO analysis for biological processes is shown in the following example:
5. Another widely-used tool for bioinformatic analysis is KEGG (Kyoto Enclycopedia for Genes and Genomes) Pathway Analysis, which we use to group positive antibody-antigen interactions on the basis of defined biologica pathways, as displayed in the example below:
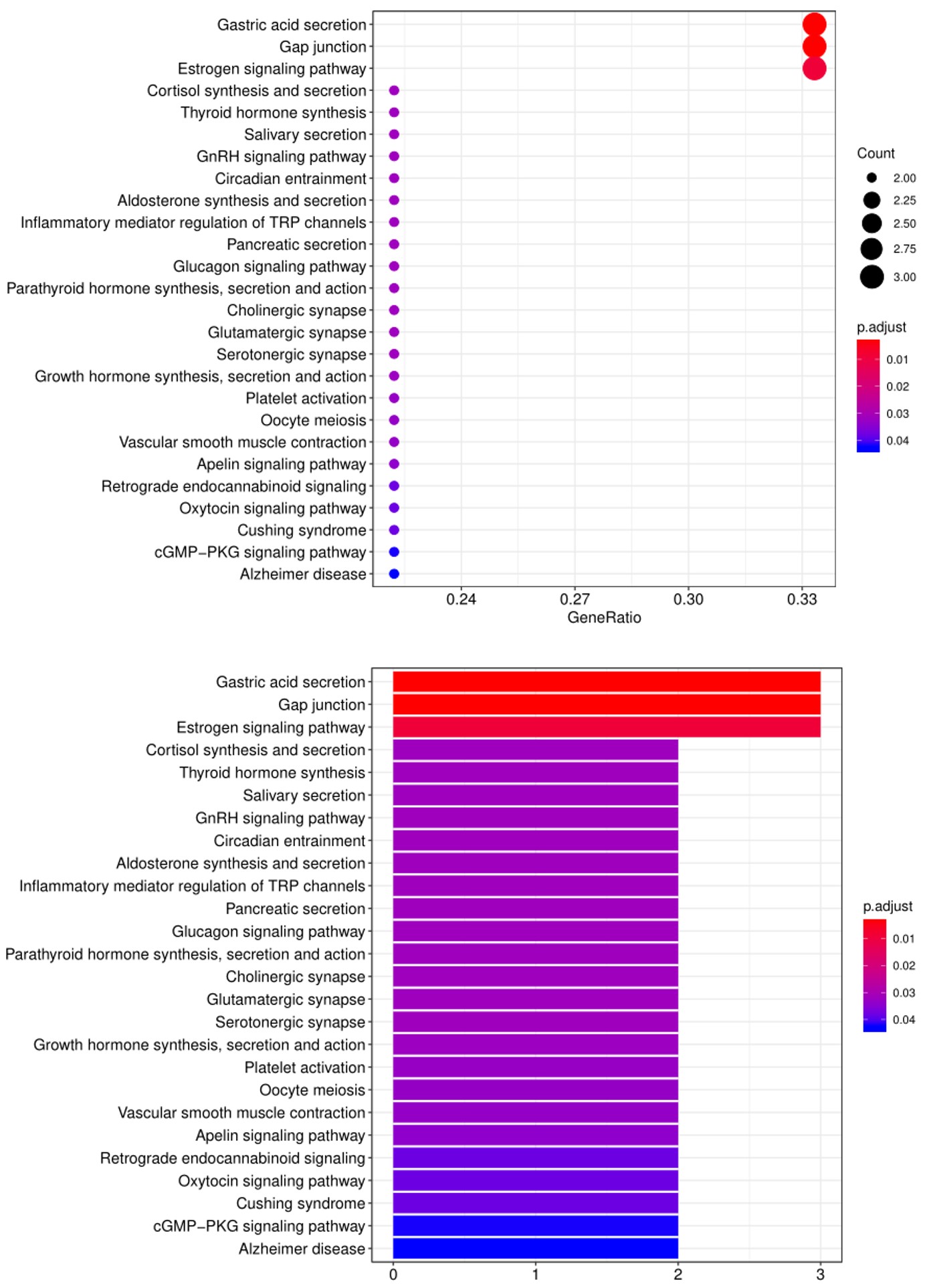
FAQ
Frequently Asked Questions
Answer: You can send us virtually any biological sample, including whole blood, serum, plasma, interstitial fluid, semen, urine, and saliva. To learn more, please contact us at
inquiry@genecopoeia.com.
Answer: Yes.
Answer: Yes.
Answer: Yes.
Answer: Each nitrocellulose membrane is surrounded by a gasket to prevent leakage and cross-contamination.
Answer: a. Each slide carries 16 identical arrays. One of these is reserved for a PBS control, while the remaining 15 are used for incubation of each sample. Therefore, the total number of samples sent by the customer is preferably a multiple of 15. However, if you have fewer than 15 samples, we will need to charge you for the full cost of an entire slide of 15 samples. b. Theoretically, each of the different test groups requires a minimum of 3 samples to meet statistical reproducibility requirements. On this basis, the greater the number of samples, the higher the reproducibility of the results in the test group. In order to ensure the reliability of the results, the sample size of different test groups is recommended to be ≥30.
Answer: The antigen microarray uses fluorescence detection, and its sensitivity is higher and more stable than ELISA (colorimetry) and Western (chemiluminescence). The technical repetition correlation coefficient R2 between the arrays can reach 0.9 or more. The dynamic detection range is 1-65000. The larger the dynamic range, the more layers of signals that can be detected.
Answer: Please fully communicate with our Technical Support staff before the experiment to explain the purpose of the experiment and the sample status, to determine whether we can meet your needs and determine the experimental plan.
Answer: The antigens on the antigen array are all derived from self-antigens reported in the literature. For each of the different types of antigenic arrays, we have shown 20 related antigens. After you have signed the appropriate service agreement, we will provide you with all the antigens and related information before you publish your own antigen array test results. Please do not disclose this information to third parties.
Answer: Proteins are usually expressed and purified from E. coli or mammalian cells.
Answer: We usually spot the entire full-length protein in its native conformation. However, we can also spot truncated proteins or peptides if needed.
Answer: If you need a custom antigen microarray, we need to fully communicate with you about the following issues: a. The intellectual property of the antigen (whether from a published paper or a patent, etc.). We can only provide customized detection of antigens that do not involve patent protection; b. Basic information about the antigen (whether it is whole protein or peptide, molecular weight, domain, whether it is a membrane protein, etc.); c. Whether you can provide an antigen that meets the requirements of a custom array; d. If we need to provide the antigen to be tested, the corresponding cost and experimental time frame need to be accounted for separately.
Answer: The antigens on our predesigned arrays are usually human. However, for custom arrays, we can use antigens from virtually any organism.In addition, the arrays have been used with human, mouse, and rat samples.
Answer: Antigens are usually spotted at a concentration between 0.1 and 1.0 mg/ml, but each individual protein concentration requires optimization.
Answer: a. This difference may be related to the detection method. The protein used on the array is a non-denatured protein, which is different from western blot. The hybrid system of the chip is an antigen-trapping antibody, and the western blot antigen is slightly different from the antibody after being transfected with the antigen. Therefore, it may cause some differences; b. Please provide a graph of the results of western blot verification, so that we can better analyze the image and data of your verified indicators on the array.








